Antonio Jimenez-Marin, Diego Rivera, Victoria Boado, Ibai Diez, Fermin Labayen, Irati Garrido, Daniela Ramos-Usuga, Javier Rasero, Alberto Cabrera-Zubizarreta, Iñigo Gabilondo, Sebastiano Stramaglia, Juan Carlos Arango Lasprilla, Jesus M. Cortes. Resting State Analysis in Multiorgan Failure Reveals Default Mode Network hyperconnectivity. OHBM 2019 – Organization for Human Brain Mapping [pdf]
Introduction:
Multiorgan Failure (MF) is a life-threating and potentially reversible condition that can affect patients admitted to Intensive Care Units (ICUs). It has been noted that patients who survive MF with no apparent brain damage (NABD) frequently present long-term neurological, cognitive and neuropsychiatric defects [Ehlenbach, 2010; Iwashyna et al., 2010; Latronico and Bolton, 2011; Pandharipande et al., 2013; Wolters et al., 2013]. However, the changes to the brain that explain such disabilities are unclear. Here, we performed a cross-sectional study and assessed patient-control groups in 4 cognitive domains together with functional MRI.
Methods:
This work was approved by the Ethics Committee at the Cruces University Hospital (Principal Investigator: Jesus M Cortes). A population of N=13 (8 males) MF patients, aged between 31 and 64 (51.54±9.36) and N=13 (6 males) healthy controls, aged between 29 and 65 (52.92±10.17), provided information consent forms. All participants underwent a complete neuropsychological evaluation and a 3T brain magnetic resonance imaging (MRI) session on the same day. For the neuropsychological evaluation, we assessed quality of life (QoL) with the 36-Item Short Form Survey (SF-36), depressive symptoms with the Patient Health Questionnaire (PHQ-9) and overall cognition with the MMSE. In addition, we performed a comprehensive neuropsychological assessment of the subjects using a battery of tests to assess the following four cognitive domains: 1) Learning and memory, 2) Speed of information processing and attention, 3) Executive functioning and 4) Language.
For the imaging analysis, we acquired 3D Turbo Field Echo (TFE) T1 images and resting state functional MRI images (rs-fMRI). With the MRI images and using a functional atlas of 2754 regions of interest (ROIs) obtained following an unsupervised clustering method [Craddock et al., 2013], we got the functional connectivity (FC) matrix of each patient. We also created seven different standard resting state networks (RSNs: default mode, cerebellum, executive control, fronto-parietal, sensorimotor, auditory and visual networks) [Smith et al., 2009]. Brain maps of strength were obtained by summing the FC matrix over either rows or columns. In addition to the strength maps obtained from the all-to-all FC matrix, maps from alternative FC matrices were built by calculating the correlations of all ROIs in the brain to those ROIs belonging only to each speci c RSN mask, thereby obtaining rectangular rather than square FC matrices of dimension 2754 x[RSN Size]. Based on the signal-detection theory, we evaluated the separability of the connectivity strength matrices for the MF and HC groups using the signal to noise ratio (SNR). Therefore, the higher the SNR, the larger the separability of the two distributions.
Results:
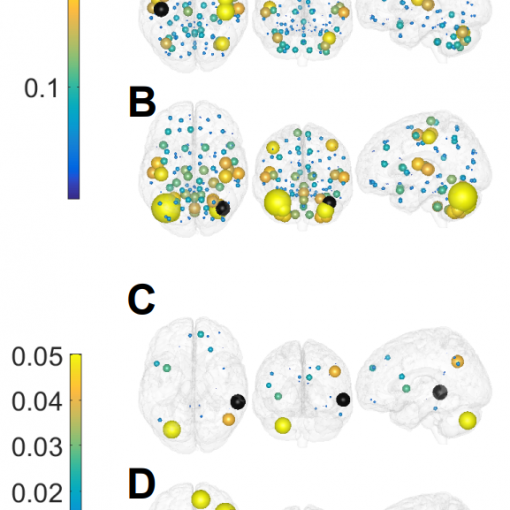
The clinical and neuropsychological variables for MF were compared with those for the HCs, MF patients generally performed worse in all cognitive domains, although no signi cant differences existed for any of the tests. Moreover, the QoL of MF patients was signi cantly worse than that of the HCs. In the imaging analysis, the only situation that produced signi cant group differences was the contrast [1 -1] (MF>HC) for the all-to-DMN, with no other FC matrix producing group differences. This DMN hyperconnectivity was found majorly to primary sensory networks but also to multimodal integration networks and higher cognitive networks. Moreover, we found that the DMN hyperconnectivity in MF patients was signi cantly and negatively correlated with performance in the ROFC Copy test ( rst domain) and SDMT test (second domain) and QoL.
Conclusions:
We found rs-fMRI changes in the MF patients compared to HCs, but no signi cant differences were found in the neuropsychological outcome. Future studies will prove if these changes in MF patient’s brain will last at long term thus anticipating the neuropsychological differences, consolidating DMN hyperconnectivity as a biomarker in MF disease.